|
|
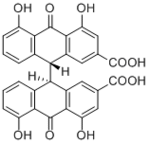
| Compound | Sennidin A |
| Animal species | rat intestinal bacteria |
| Metabolism parameters | |
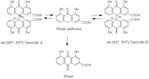
| Metabolites |
rel-(9R*, 9R*)-Sennidin A
Rhein anthrone rel-(9R*, 9R*)-Sennidin B Rhein |
| Crude drug | Rhubarb , Senna Leaf |
| References | 1) Hattori M., Kim G., Motoike S., Kobashi K. and Namba T.: Metabolism of sennosides by intestinal flora. Chem. Pharm. Bull., 30, 1338-1346 (1982). 2) Akao T., Mibu K., Erabi T., Hattori M., Namba T. and Kobashi K.: Non-enzymatic reduction of sennidins and sennosides by reduced flavin. Chem. Pharm. Bull., 35, 1998-2003 (1987). |
| Remarks | ※Preparation of a suspension of rat feces and its supernatant fluid Fresh feces (20 g) of Wistar rats (female, 180-220 g body weight) were suspended in 100 mM phosphate buffer (200 ml, pH 7.3) containing 0.05% cysteine, which had previously been bubbled through with carbon dioxide to eliminate air. The supernatant fluid was prepared by centrifuging the suspension at 10000 rpm for 10 min. [Hattori et al., Chem. Pharm. Bull., 30, 1338-1346 (1982)] ※Non-enzymatic reduction of sennidins, sennosides and methyl orange Non-enzymatic reduction of these compounds was carried out in a Thunberg-type tube under nitrogen. In the main part of the tube, 0.2-20 μmol of a cofactor such as FAD, FMN, riboflavin or benzyl viologen was first reduced with 4-100 μmol of NADH in the presence of the purified sennidin reductase in 2 ml of 0.1 m K-phosphate buffer at 37 °C. Then 0.19 μmol of sennidin A or B, 0.23 μmol of sennoside A or B, or 0.20 μmol of methyl orange containing 2 μmol of p-chloromercuriphenylsulfonic acid (pCMS) (to inactivate the enzyme) in the side arm was mixed with the reduced cofactor in the main tube at room temperature. Rhein anthrone and 8-glucosylrhein anthrone, the reduction products of sennidins and sennosides, respectively, were determined as the azometin derivatives by adding p-nitoroso-N,N'-dimethylaniline. Reduced methyl orange was determined by measuring the absorbance at 500 nm. [Akao et al., Chem. Pharm. Bull., 35, 1998-2003 (1987)] |

